Functional Genomics of Hemoglobin Switching
Despite the fundamental importance of BCL11A’s role in HbF regulation, much of hemoglobin switching remains poorly understood. Critical regulators of fetal hemoglobin level remain to be discovered. First, the upstream signaling pathways activating BCL11A in adult-stage erythroid cells are largely unknown. Second, the cofactors interacting with BCL11A only explain a fraction of BCL11A’s effects, implying additional cooperating factors. Finally, even in the absence of BCL11A, hemoglobin switching still occurs (albeit at reduced amplitude), suggesting that independent pathways contribute to γ-globin repression in adult erythropoiesis. We have conducted a genome-scale pooled shRNA screen in primary human erythroid precursors to discover novel regulators of HbF. We are using CRISPR/ Cas9 for orthogonal discovery and validation. Current projects include conducting mechanistic studies of individual candidates. These studies are expected to identify novel regulators and therapeutic targets that contribute to hemoglobin switching upstream, downstream, and parallel to BCL11A.
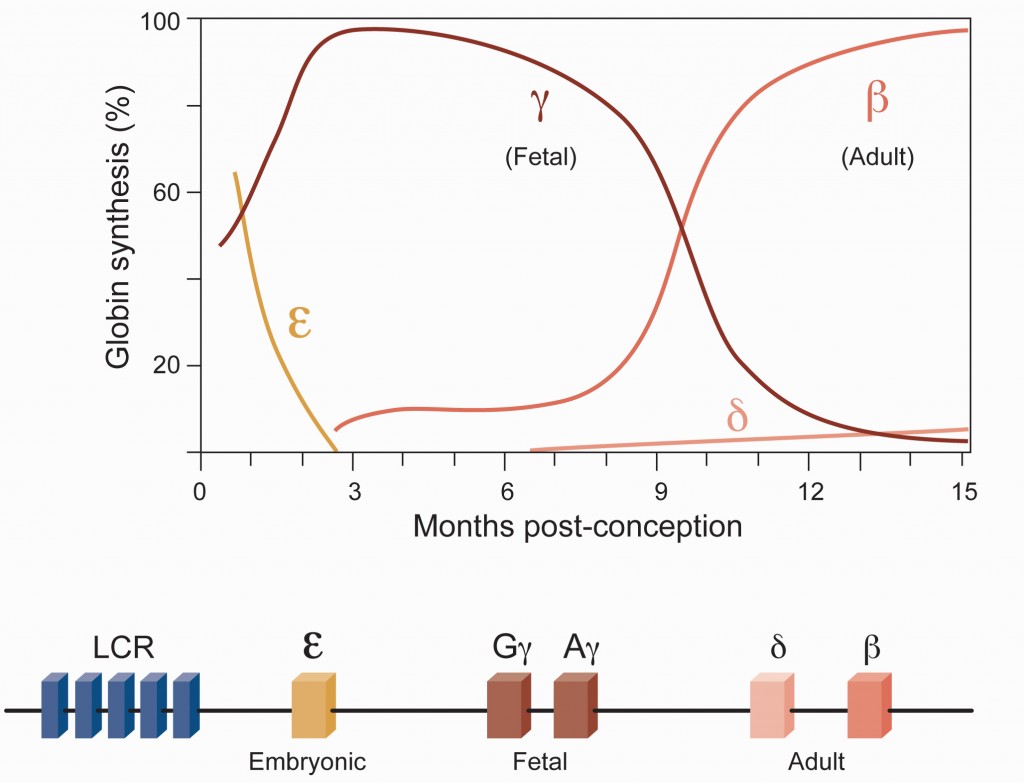
Image: Hemoglobin switching refers to the changes in globin gene expression during ontogeny. The switch from [gamma]- to [beta]-globin expression is of particular clinical importance for the [beta]-globin disorders. Adapted from Bauer, Kamran, Orkin, 2013.
